Pyocyanin (Synonyms: Sanasin,Sanazin,Pyocyanine) |
| Catalog No.GC13137 |
Pyocyanin is a biologically active phenazine pigment
Products are for research use only. Not for human use. We do not sell to patients.
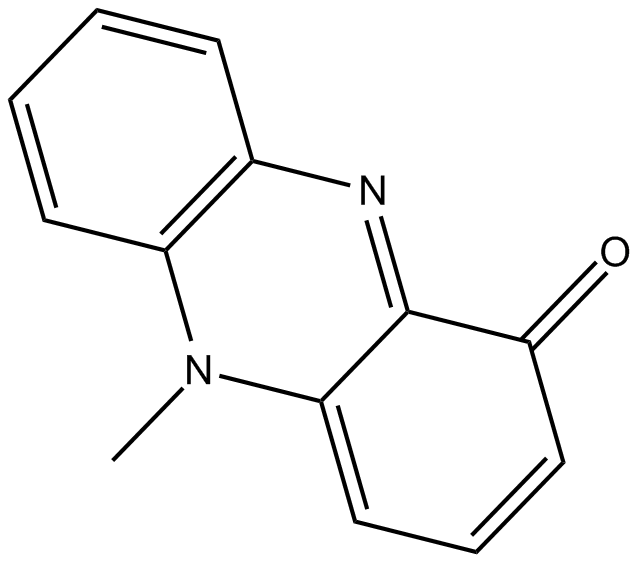
Cas No.: 85-66-5
Sample solution is provided at 25 µL, 10mM.
Pyocyanin is a biologically active phenazine pigment produced by the bacterium, Pseudomonas aeruginosa, acting as a nitric oxide (NO) antagonist in various pharmacological preparations and as mediator in biosensors. Pyocyanin can also be used as electron shuttle in microbial fuel cells enabling bacterial electron transfers. Furthermore, Pyocyanin has broad antibiotic activity as well as has been identified as the key molecule produced by Pseudomonas that inhibits growth of pathogenic vibrios in aquaculture systems.[1]
In vitro study was performed to measure the cytotoxicity of Pyocyanin. Results indicated that L-132 cells were prone to Pyocyanin-induced toxicity. The IC50 value of Pyocyanin on inhibition of mitochondrial dehydrogenase activity was 112.01 ± 23.73 mg l−1. The IC50 value of Pyocyanin induced damage of plasma membrane was 21.79 ± 14.23 mg l−1. Moreover, Pyocyanin showed an IC50 of 32.57 ± 16.52 mg l−1 on inhibition of protein synthesis. When Pyocyanin has concentration of 25 mg l−1, 3.9 % inhibition of mitochondrial activity, 47.3 % plasma membrane damage and 26.6 % inhibition of protein synthesis were observed in L-132 cells. Whereas at lower concentration (6.25 mg l−1) the toxicity was negligible, whereas at 200 mg l−1 the values were 64.8, 72.8 and 91.7 %, respectively.[1]
In vivo study demonstrated that Pyocyanin was able to slow the beating of human respiratory tract cilia. The effects of Pyocyanin on tracheal mucus velocity of radiolabeled erythrocytes were tested in anesthetized guinea pigs. The effect of Pyocyanin was slower in onset, 600 ng causing 60% reduction in tracheal mucus velocity at 3 h, and no recovery occurred. Whereas combination of Pyocyanin and 1-hydroxyphenazine produced an initial rapid slowing equivalent to the same dose of 1-hydroxyphenazine given alone, but the later slowing attributed to Pyocyanin was greater than the same dose administered alone. This study demonstrates one mechanism by which products of P. aeruginosa, such as Pyocyanin may facilitate its colonization of the respiratory tract.
References:
[1]. Priyaja P, et al. Pyocyanin induced in vitro oxidative damage and its toxicity level in human, fish and insect cell lines for its selective biological applications. Cytotechnology. 2016 Jan;68(1):143-155.
[2]. Arora D, et al. Pyocyanin induces systemic oxidative stress, inflammation and behavioral changes in vivo. Toxicol Mech Methods. 2018 Jul;28(6):410-414.
Average Rating: 5 (Based on Reviews and 18 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




