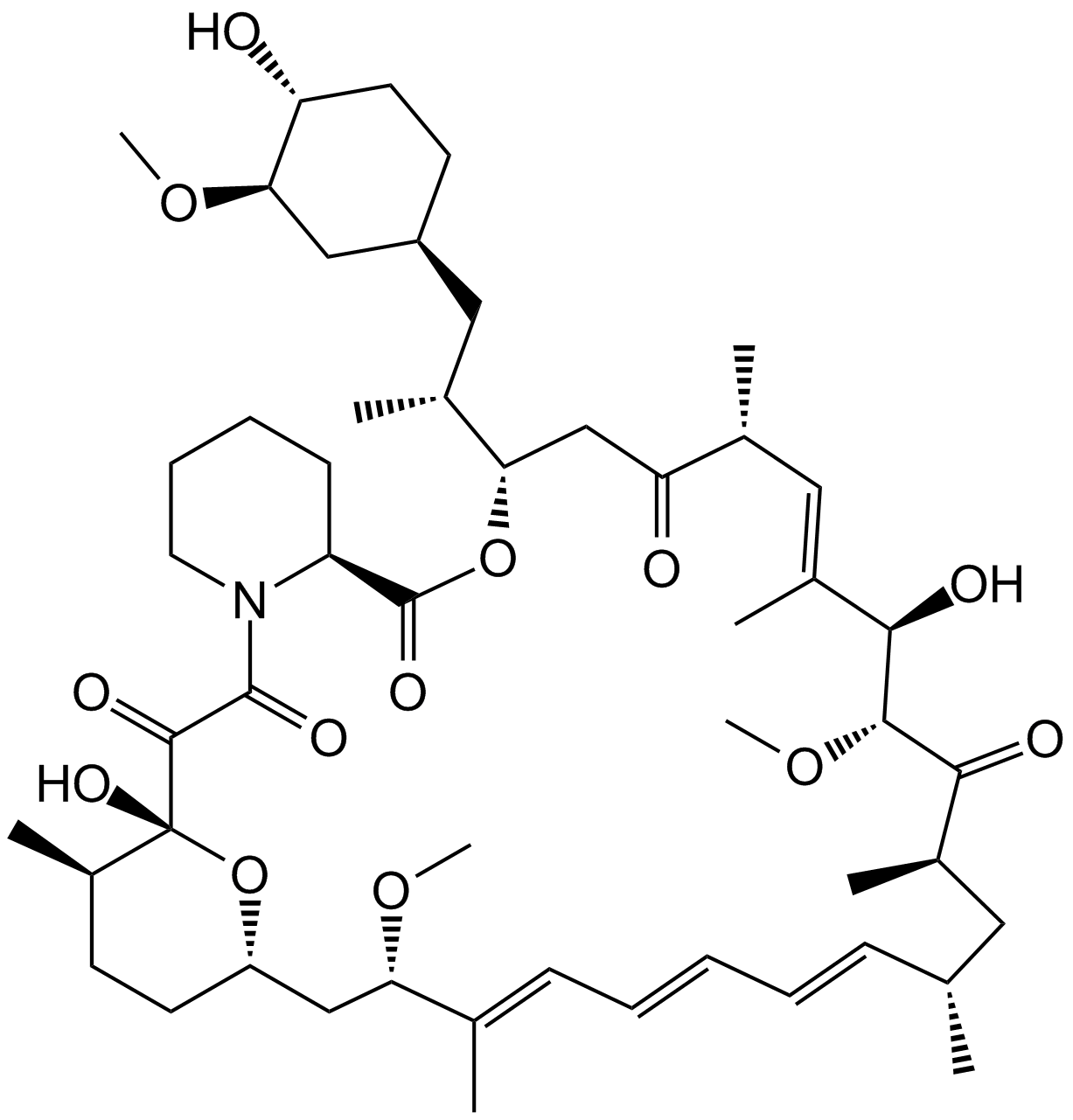
Rapamycin (Sirolimus) (Synonyms: AY 22989, NSC 226080, Sirolimus, Wy 090217) |
| Catalog No.GC15031 |
Rapamycin used to be used as an antifungal antibiotic.
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 53123-88-9
Sample solution is provided at 25 µL, 10mM.
Rapamycin used to be used as an antifungal antibiotic[3]. Rapamycin exerts immunosuppressive effects by inhibiting the activation and proliferation of T cells. Rapamycin binds to FK-binding protein 12 (FKBP12) to form the Rapamycin-FKBP12 complex, which can inhibit mTOR[4,6].As a potent and specific mTOR inhibitor with an IC50?of 0.1 nM in HEK293 cells. Rapamycin binds to FKBP12 and specifically acts as an allosteric inhibitor of mTORC1[5].
Rapamycin (12.5-100 nM; 24 hours) treatment exerts modest inhibitory effect on lung cancer cell proliferation in a dose-dependent manner in all cell lines (A549, SPC-A-1, 95D and NCI-H446 cells) tested, achieving about 30-40% reduction in cell proliferation at 100 nM vs. ~10% reduction at 12.5 nM[7].Rapamycin potently not only suppressed proliferation but also induced the apoptosis of LECs in a dose-dependent manner under HGF administration. Rapamycin could promote apoptosis of LECs via inhibiting HGF-induced phosphorylation of AKT/mTOR, ERK and JAK2/STAT3 signaling molecules[1].
Rapamycin reduces neurologic disease in Ndufs4 -- / -- mice. In rapamycin treated knockout mice, the percentage of mice exhibiting neurological symptoms was greatly reduced at each age point after P35, and about half of these mice never showed obvious signs of neurological disease before dying[2]. Rapamycin alone has a moderate inhibitory effect. However, the combination of Metformin and Rapamycin exerts a significantly increased inhibition of tumor growth compared with the control group, the Rapamycin monotherapy group and the Metformin monotherapy group[8].Rapamycin treatment in cell culture significantly inhibits c-Myc-regulated gene expression. Rapamycin suppresses tumor growth along with a decreased expression of STAT3 and c-Myc in an in vivo xenograft mouse model for hepatocellular carcinoma[9].
References:
[1]: Tian F, Dong L, et,al. Rapamycin-Induced apoptosis in HGF-stimulated lens epithelial cells by AKT/mTOR, ERK and JAK2/STAT3 pathways. Int J Mol Sci. 2014 Aug 11;15(8):13833-48. doi: 10.3390/ijms150813833. PMID: 25116684; PMCID: PMC4159827.
[2]: Johnson SC, Yanos ME, et,al. mTOR inhibition alleviates mitochondrial disease in a mouse model of Leigh syndrome. Science. 2013 Dec 20;342(6165):1524-8. doi: 10.1126/science.1244360. Epub 2013 Nov 14. PMID: 24231806; PMCID: PMC4055856.
[3]: Sehgal SN, Baker H, et,al. Rapamycin (AY-22,989), a new antifungal antibiotic. II. Fermentation, isolation and characterization. J Antibiot (Tokyo). 1975 Oct;28(10):727-32. doi: 10.7164/antibiotics.28.727. PMID: 1102509.
[4]: Sehgal SN. Rapamune (RAPA, rapamycin, sirolimus): mechanism of action immunosuppressive effect results from blockade of signal transduction and inhibition of cell cycle progression. Clin Biochem. 1998 Jul;31(5):335-40. doi: 10.1016/s0009-9120(98)00045-9. PMID: 9721431.
[5]: Edwards SR, Wandless TJ. The rapamycin-binding domain of the protein kinase mammalian target of rapamycin is a destabilizing domain. J Biol Chem. 2007 May 4;282(18):13395-401. doi: 10.1074/jbc.M700498200. Epub 2007 Mar 9. PMID: 17350953; PMCID: PMC3763840.
[6]: Rangaraju S, Verrier JD, et,al. Rapamycin activates autophagy and improves myelination in explant cultures from neuropathic mice. J Neurosci. 2010 Aug 25;30(34):11388-97. doi: 10.1523/JNEUROSCI.1356-10.2010. PMID: 20739560; PMCID: PMC3478092.
[7]: Niu H, Wang J, et,al. Rapamycin potentiates cytotoxicity by docetaxel possibly through downregulation of Survivin in lung cancer cells. J Exp Clin Cancer Res. 2011 Mar 10;30(1):28. doi: 10.1186/1756-9966-30-28. PMID: 21392382; PMCID: PMC3065416.
[8]: Zhang JW, Zhao F, et,al. Metformin synergizes with rapamycin to inhibit the growth of pancreatic cancer in vitro and in vivo. Oncol Lett. 2018 Feb;15(2):1811-1816. doi: 10.3892/ol.2017.7444. Epub 2017 Nov 20. PMID: 29434877; PMCID: PMC5774390.
[9]: Sun L, Yan Y, et,al. Rapamycin targets STAT3 and impacts c-Myc to suppress tumor growth. Cell Chem Biol. 2022 Mar 17;29(3):373-385.e6. doi: 10.1016/j.chembiol.2021.10.006. Epub 2021 Oct 26. PMID: 34706270.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *










