Actagardin (Synonyms: Antibiotic A 3802-IV-3,Gardimycin) |
| Catalog No.GC10552 |
tetracyclic antibiotic
Products are for research use only. Not for human use. We do not sell to patients.
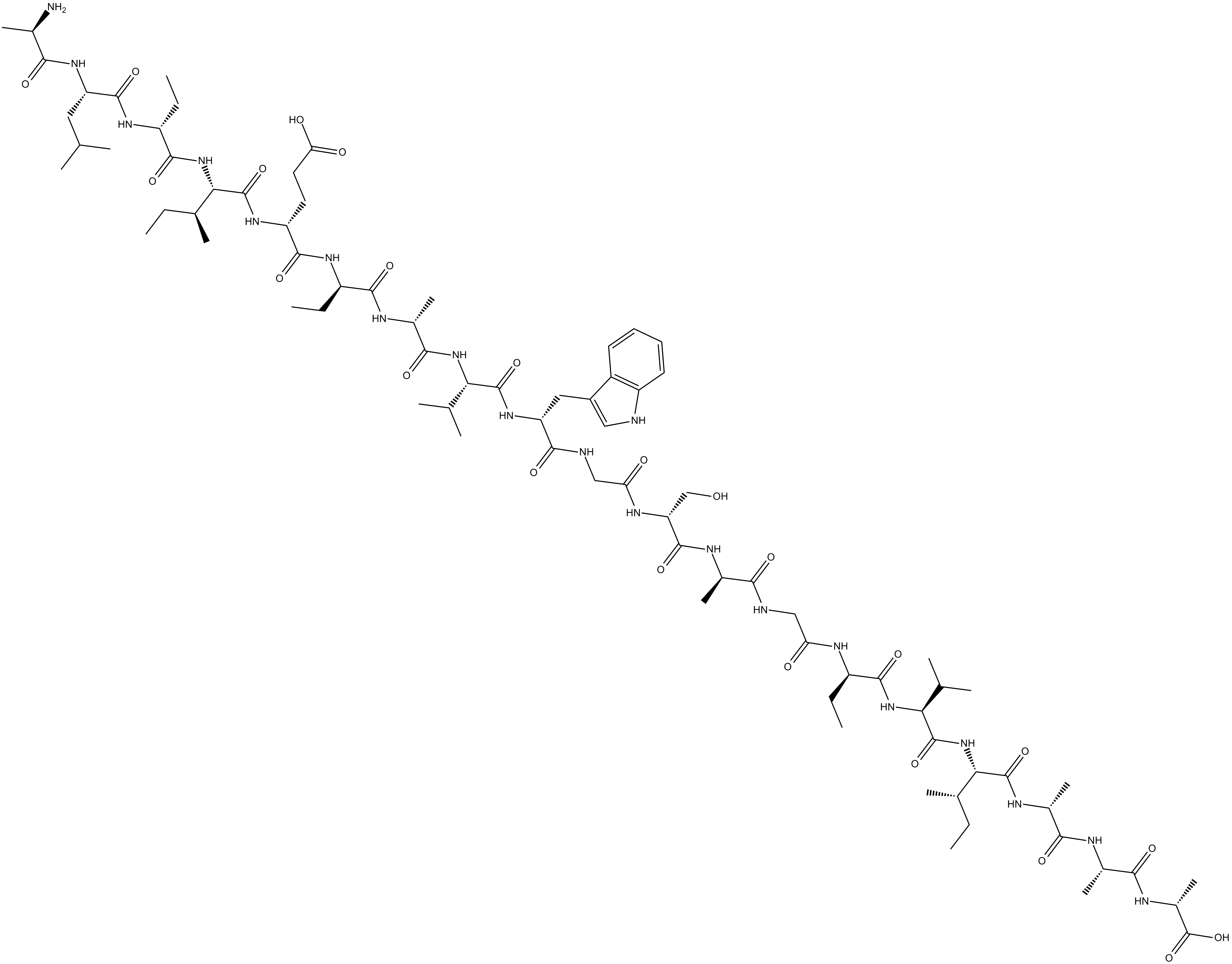
Cas No.: 59165-34-3
Sample solution is provided at 25 µL, 10mM.
Actagardin is a tetracyclic antibiotic.
Actagardin is a tetracyclic antibiotic produced by several species of Actinoplanes.
In vitro: During the repeated fermentations of actagardin, two new compounds, named D and E, were isolated. Compound D was found to be twice as active as actagardin against Streptococcus pyogenes C 203 and four times more active than actagardin against Staphylococcus aureus ATCC 6538, S. pneumoniae UC 41, and S. aureus TOUR in the in-vitro tests [1].
In vivo: Animal study showed that compound D was also more effective than the parent compound actagardin against experimental infections in mice with either S. pyogenes C 203 or S. aureus TOUR, after sc administration. However, compound D was ineffective at doses up to 150 mg/kg when given orally to mice infected with S. pyogenes C 203. Moreover, compound D (LD50 1,250 mg/kg, mice, ip) was about 2.5 times more toxic than actagardine (3,310 mg/kg). In addition, compound E showed practically no in vitro activity up to 50 μg/ml against the tested organisms [1].
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] Malabarba A, Landi M, Pallanza R, Cavalleri B. Physico-chemical and biological properties of actagardine and some acid hydrolysis products. J Antibiot (Tokyo). 1985 Nov;38(11):1506-11.
Average Rating: 5 (Based on Reviews and 3 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















