Amitriptyline HCl (Synonyms: NIH 10794, Ro 4-1575) |
| Catalog No.GC13723 |
Amitriptyline HCl is an inhibitor of serotonin reuptake transporter (SERT) and noradrenaline reuptake transporter (NET), with Kis of 3.45 nM and 13.3 nM for human SERT and NET, respectively.
Products are for research use only. Not for human use. We do not sell to patients.
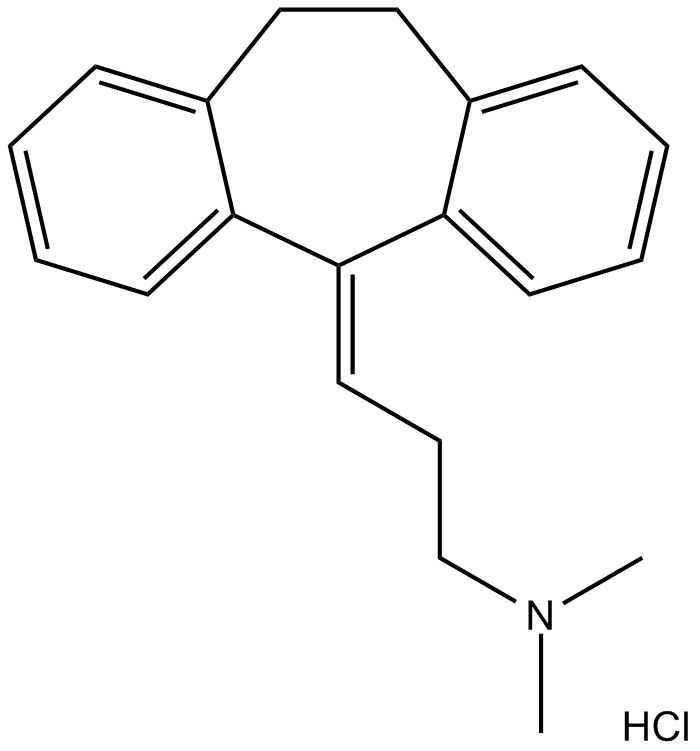
Cas No.: 549-18-8
Sample solution is provided at 25 µL, 10mM.
Amitriptyline hydrochloride is a dibenzocycloheptene-derivative tricyclic antidepressant (TCA).Target: OthersAmitriptyline hydrochloride acts primarily as a serotonin-norepinephrine reuptake inhibitor, with strong actions on the serotonin transporter and moderate effects on the norepinephrine transporter. It has negligible influence on the dopamine transporter and therefore does not affect dopamine reuptake, being nearly 1,000 times weaker on it than on serotonin [1]. Amitriptyline additionally functions as a 5-HT2A, 5-HT2C, 5-HT3, 5-HT6, 5-HT7, α1-adrenergic, H1, H2, and mACh receptorantagonist, and σ1 receptor agonist. It has also been shown to be a relatively weak NMDA receptor negative allosteric modulator at the same binding site as phencyclidine. Amitriptyline hydrochloride inhibits sodium channels, L-type calcium channels, and Kv1.1, Kv7.2, and Kv7.3 voltage-gated potassium channels, and therefore acts as a sodium, calcium, and potassium channel blocker as well [2]. Recently, amitriptyline has been demonstrated to act as an agonist of the TrkA and TrkB receptors. It promotes the heterodimerization of these proteins in the absence of NGF and has potent neurotrophic activity both in-vivo and in-vitro in mouse models [3].
References:
[1]. Tatsumi, M., et al., Pharmacological profile of antidepressants and related compounds at human monoamine transporters. Eur J Pharmacol, 1997. 340(2-3): p. 249-58.
[2]. http://en.wikipedia.org/wiki/Amitriptyline
[3]. Jang, S.W., et al., Amitriptyline is a TrkA and TrkB receptor agonist that promotes TrkA/TrkB heterodimerization and has potent neurotrophic activity. Chem Biol, 2009. 16(6): p. 644-56.
Average Rating: 5 (Based on Reviews and 13 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




