Bivalirudin TFA |
| Catalog No.GC35528 |
Bivalirudin TFA is a synthetic 20 residue peptide which reversibly inhibits thrombin.
Products are for research use only. Not for human use. We do not sell to patients.
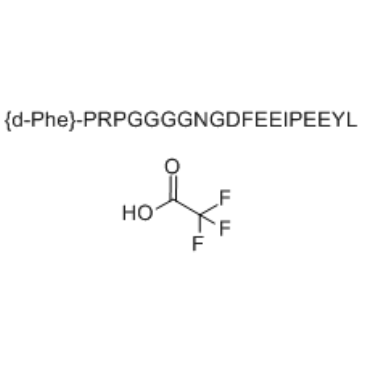
Cas No.: 1191386-55-6
Sample solution is provided at 25 µL, 10mM.
Bivalirudin TFA is a synthetic 20 residue peptide which reversibly inhibits thrombin.IC50 Value:Target: thrombinin vitro: Eptifibatide (8 mg/mL) added together with a low (70 ng/mL) concentration of bivalirudin (a direct thrombin inhibitor) effectively (approximately 90%) reduced platelet aggregation induced by thrombin (0.2 U/mL) [1]. In thrombin generation assay (TGA), bivalirudin had no effect on these parameters up to 10 μmol/l [2]. Bivalirudin-facilitated binding of MPO to BAEC resulted also in functional changes in terms of increased NO consumption as well as enhanced MPO-mediated redox modifications [3].in vivo: The use of bivalirudinprevented further increase in antiheparin/PF4 antibody IgG levels in rats [4]. Three animals in the 500-mg/kg/24 h group, and 7 animals in the 2000-mg/kg/24 h group in the toxicokinetic assessment phase of the study were found dead or euthanized in extremis (following blood sampling). Plasma concentrations of bivalirudin appeared to be linear and dose independent [5].Clinical trial: Antithrombotic Effects of Ticagrelor Versus Clopidogrel . Phase 4
[1]. Ciborowski M, Tomasiak M. The in vitro effect of eptifibatide, a glycoprotein IIb/IIIa antagonist, on various responses of porcine blood platelets. Acta Pol Pharm. 2009 May-Jun;66(3):235-42. [2]. Xu Y, Wu W, Wang L, Differential profiles of thrombin inhibitors (heparin, hirudin, bivalirudin, and dabigatran) in the thrombin generation assay and thromboelastography in vitro. Blood Coagul Fibrinolysis. 2013 Apr;24(3):332-8. [3]. Rudolph V, Rudolph TK, Schopfer FJ, Bivalirudin decreases NO bioavailability by vascular immobilization of myeloperoxidase. J Pharmacol Exp Ther. 2008 Nov;327(2):324-31. [4]. Zhang R, Huang Y, Zhang M, Bivalirudin Utilization in Rats Undergoing Cardiopulmonary Bypass: Preventing the Increase of Antiheparin/Platelet Factor 4 Antibody in Perioperative Period. Clin Appl Thromb Hemost. 2012 Aug 21. [Epub ahead of print] [5]. Gleason TG, Chengelis CP, Jackson CB, A 24-hour continuous infusion study of bivalirudin in the rat. Int J Toxicol. 2003 May-Jun;22(3):195-206.
Average Rating: 5 (Based on Reviews and 9 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















