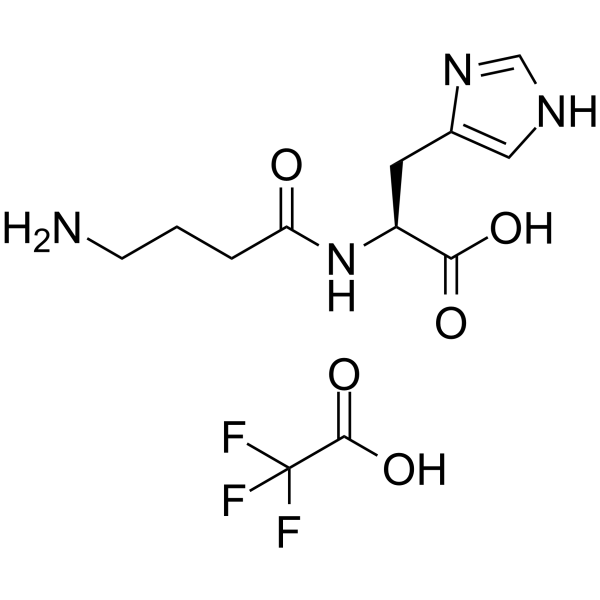
Homocarnosine TFA (Synonyms: L-Homocarnosine TFA; γ-Aminobutyryl-L-histidine TFA) |
| Catalog No.GC63950 |
Homocarnosine TFA is a dipeptide of γ-aminobutyric acid (GABA) and histidine unique to brain.
Products are for research use only. Not for human use. We do not sell to patients.
Sample solution is provided at 25 µL, 10mM.
Homocarnosine TFA is a dipeptide of γ-aminobutyric acid (GABA) and histidine unique to brain. Homocarnosine TFA is an inhibitory neuromodulator synthesized in the neuron from GABA and exhibiting anticonvulsant effects[1]. Homocarnosine TFA has antioxidant and anti-inflammatory actions, prevention of DNA damage, and inhibition of advanced glycation end-product formation[2].
Homocarnosine, a dipeptide of γ-aminobutyric acid (GABA) and histidine, is an inhibitory neuromodulator synthesized in subclasses of GABAergic neurons[1].Homocarnosine is a potent alternative imidazole peptide for skeletal muscle based on its structural similarity to Carnosine. Homocarnosine is composed of γ-aminobutyric acid (GABA) and histidine and has a similar structure to Carnosine, with the only difference being an extra carbon atom in GABA compared to that in β-alanine of carnosine. Homocarnosine possesses greater tolerance to degradation by serum carnosinase relative to Carnosine. Homocarnosine is known to be exclusively present in brain and cerebrospinal fluid and acts as a neuroprotective agent in a broad range of disease conditions. Homocarnosine exerts antioxidant activity and prevent DNA oxidative damage to a similar extent as Carnosine[2].
[1]. Petroff OA, et al. Vigabatrin increases human brain homocarnosine and improves seizure control. Ann Neurol. 1998;44(6):948-952.
[2]. Kumrungsee T, et al. Dietary GABA induces endogenous synthesis of a novel imidazole peptide homocarnosine in mouse skeletal muscles. Amino Acids. 2020;52(5):743-753.
Average Rating: 5 (Based on Reviews and 3 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















