ITX5061 |
| Catalog No.GC32209 |
ITX5061 is a type II inhibitor of p38 MAPK and also an antagonist of scavenger receptor B1 (SR-B1).
Products are for research use only. Not for human use. We do not sell to patients.
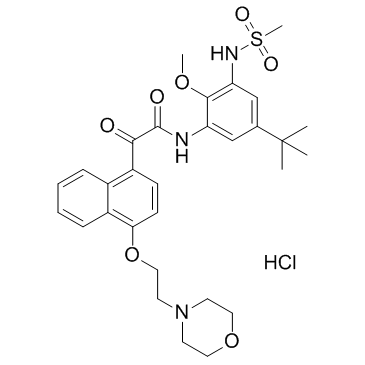
Cas No.: 1252679-52-9
Sample solution is provided at 25 µL, 10mM.
ITX5061 is a type II inhibitor of p38 MAPK and also an antagonist of scavenger receptor B1 (SR-B1).
ITX5061 is a type II inhibitor of p38 MAPK and also an antagonist of scavenger receptor B1 (SR-B1). Treatment of ITX5061 (30 mg/kg/day) for mice results in a 50% increase in HDL-C levels compare to baseline. ApoA-I levels are moderately (+15 %) but significantly increased in ITX5061-treated HuAITg mice, compare to mice receive vehicle. ITX5061 significantly decreases HDL-CE catabolism with an FCR of 1.86±0.40 pools/d vs 2.47±0.26 pools/d in the control group (P<0.05), while calculated production rates are identical in both groups (129±24 μg/g/d vs 129±16 μg/g/d). Moreover, accumulation of [3H] CE in the liver is significantly lower in ITX5061-treated mice indicating that increased HDL-CE levels are due to reduced uptake by the liver[1].
[1]. Masson D, et al. Increased HDL cholesterol and apoA-I in humans and mice treated with a novel SR-BI inhibitor. Arterioscler Thromb Vasc Biol. 2009 Dec;29(12):2054-60.
Average Rating: 5 (Based on Reviews and 24 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















