SB 431542
|
| Catalog No.GC11545 |
SB-431542, a small molecule inhibitor of the type I TGF-β receptor, blocks intracellular mediators of TGF-1 signaling, which leads to decreased TGF-β1–mediated proliferation, cytokines and collagen expression.
Products are for research use only. Not for human use. We do not sell to patients.
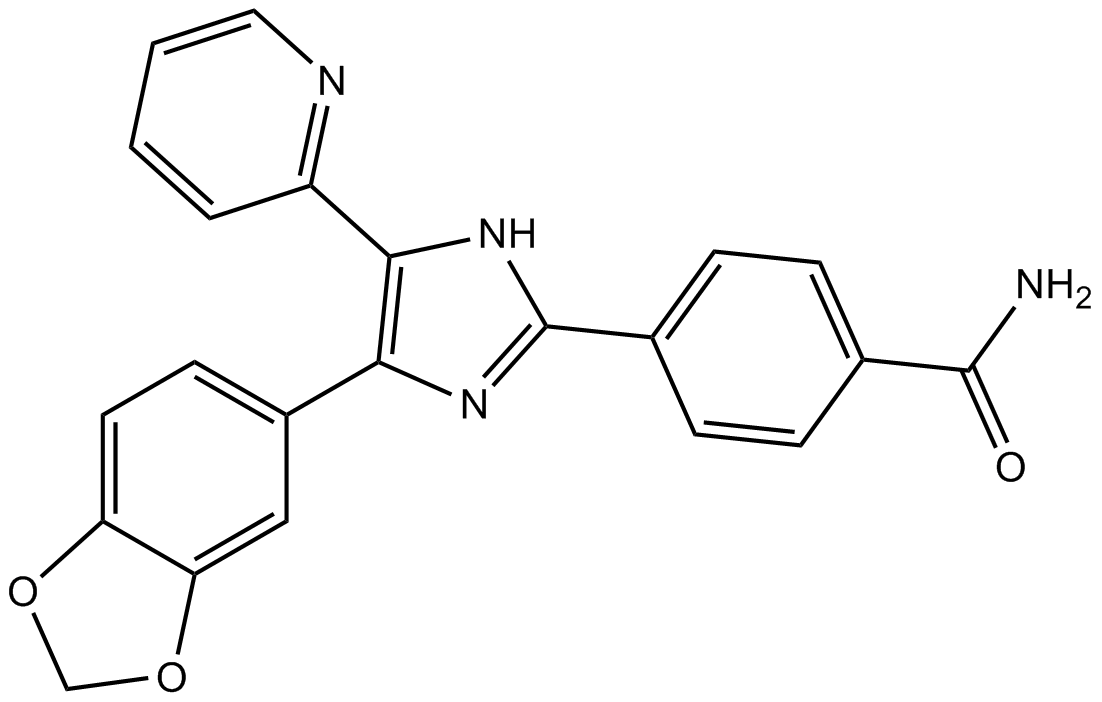
Cas No.: 301836-41-9
Sample solution is provided at 25 µL, 10mM.
SB-431542, a small molecule inhibitor of the type I TGF-β receptor, blocks intracellular mediators of TGF-1 signaling, which leads to decreased TGF-β1–mediated proliferation, cytokines and collagen expression. In clinical settings, SB-431542 is widely used to treat respiratory asthma, and inhibits proliferation and synthesis of adventitial fibro in the process of pulmonary vascular remodeling.[1]
In vitro study indicated that SB-431542 is able to inhibit ALK5 with an IC50 of 94 nM and other type I receptors, such as ALK4. Although SB-431542 inhibited ALK4 with an IC50 of 140 nM. Moreover, SB-431542 inhibited TGF-β1–induced collagen Iα1 and PAI-1 mRNA with IC50 values of 60 and 50 nM, respectively. In addition, SB-431542 inhibited TGF-β1–induced fibronectin mRNA and protein with IC50 values of 62 and 22 nM, respectively. These data demonstrate for the first time that ALK5 activity is required for TGF-β1 regulation of extracellular matrix markers FN, collagen Iα1, and PAI-1 mRNA.[1]
In vivo study demonstrated that SB-431542 has the capacity to inhibit TGF-β1-induced gene expression. SB-431542 is recognized as a important inhibitor of the TGF-β1 receptors in blocking TGF-β1/Smads signal pathways in vascular remodeling. Moreover, hypoxia-induced vascular remodeling can significantly increase the amount of cytokines and collagen in vascular adventitia. However, after the treatment of SB-431542, attenuation of the fibrosis promoting effects of TGF-β1, including TGF-β1-induced cell proliferation, cell motility, cell migration and cell synthesis were observed. Therefore, it is significant to the identify the potential of SB-431542 for the treatment of hypoxia-induced pulmonary hypertension.[2]
References:
[1]. Laping NJ, et al. Inhibition of transforming growth factor (TGF)-beta1-induced extracellular matrix with a novel inhibitor of the TGF-beta type I receptor kinase activity: SB-431542. Mol Pharmacol. 2002 Jul;62(1):58-64.
[2]. Yuan W, et al. SB-431542, a specific inhibitor of the TGF-β type I receptor inhibits hypoxia-induced proliferation of pulmonary artery adventitial fibroblasts. Pharmazie. 2016 Feb;71(2):94-100.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *






