EDP-305 |
| Catalog No.GC65260 |
EDP-305 is an orally active, potent and selective farnesoid X receptor (FXR) agonist, with EC50 values of 34 nM (chimeric FXR in CHO cells) and 8 nM (full-length FXR in HEK cells).
Products are for research use only. Not for human use. We do not sell to patients.
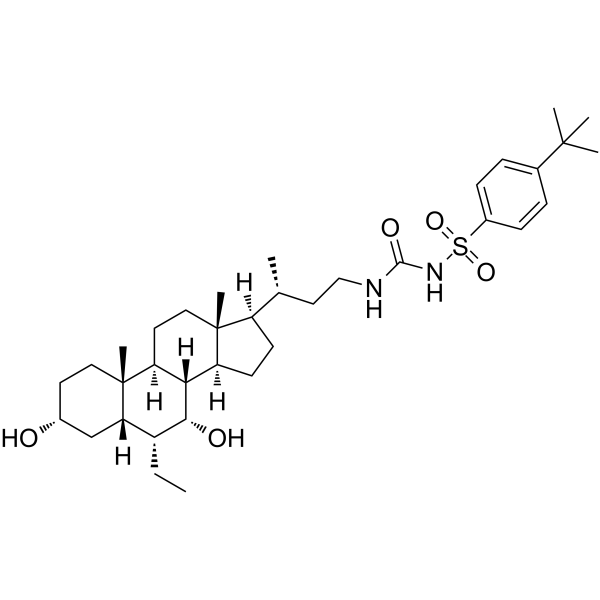
Cas No.: 1933507-63-1
Sample solution is provided at 25 µL, 10mM.
EDP-305 is an orally active, potent and selective farnesoid X receptor (FXR) agonist, with EC50 values of 34 nM (chimeric FXR in CHO cells) and 8 nM (full-length FXR in HEK cells). EDP-305 shows a potent and consistent antifibrotic effect. EDP-305 can be used for primary biliary cholangitis (PBC) and non-alcoholic steatohepatitis (NASH) research[1][2].
EDP‐305 (10 μM, 72 h) directly activates FXR in liver hepatoctyes but not stellate cells[1]. EDP-305 (0-5 μM, 16 h) increases the expression of the FXR target gene, SHP, and downregulates CYP7A1 expression in HepaRG hepatocytes[2].
EDP‐305 (0-30 mg/kg, Oral gavage, daily for 2 weeks) reduces serum markers of liver injury, and reduces liver fibrosis in a dose-dependent manner in BDL rats[1].EDP‐305 (0-30 mg/kg, Oral gavage, daily for 6 weeks) reduces liver fibrosis in a dose-dependent manner in CDAHFD mice[1].
[1]. Erstad DJ, et al. Molecular magnetic resonance imaging accurately measures the antifibrotic effect of EDP-305, a novel farnesoid X receptor agonist. Hepatol Commun. 2018 May 21;2(7):821-835. [2]. Chau M, et al. Characterization of EDP-305, a Highly Potent and Selective Farnesoid X Receptor Agonist, for the Treatment of Non-alcoholic Steatohepatitis[J]. International Journal of Gastroenterology, 2019(1).
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















