Pifithrin-α (PFTα) (Synonyms: PFTα) |
| Catalog No.GC10538 |
Pifithrin-α(PFT-α) is a p53 inhibitor. Pifithrin-αwidely used in neuroscience to block neuronal apoptotic cell death. Pifithrin-αis also a potent stimulant of aryl hydrocarbon receptor (AhR)..
Products are for research use only. Not for human use. We do not sell to patients.
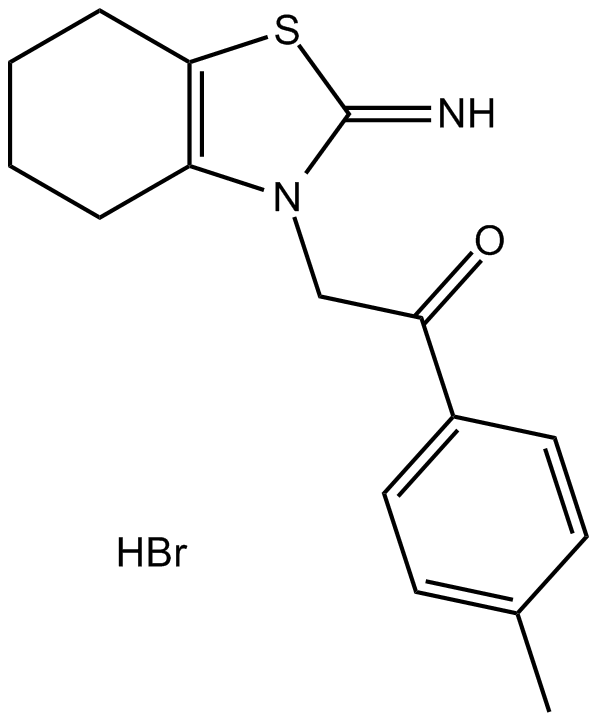
Cas No.: 63208-82-2
Sample solution is provided at 25 µL, 10mM.
Pifithrin-α(PFT-α) is a p53 inhibitor[1]. Pifithrin-α widely used in neuroscience to block neuronal apoptotic cell death[2]. Pifithrin-α is also a potent stimulant of aryl hydrocarbon receptor (AhR) [3].
Pifithrin-α (10 µM;48h) reduces meth-induced degeneration of dopaminergic neurons[4]. Treatment with Pifithrin-α(1 µM; 12-24 h) prevents ROS formation in hMp84-overexpressing A375 cells[5]. The IC50 of TPT for HepG2 cells after 10 µµ PFTα pretreated(1-100 µM; 2 h), was 4.8 to 14.4 folds lower than the effect of TPT alone. PFTα decreases the p-p53 levels and p-p53 activity, not affects p53 expression in p53 positive tumor cells[6].
Pifithrin-α (2 mg/kg; i.v) reduced the number of apoptotic cells in the ischemic brain by inhibiting the binding of p53 to its DNA sites as it reduced the expression of the p53-related gene p21WAF[7].Delayed Pifithrin-α (0.4µg/µl; 20µl; i.c.v + 0.2mg/100gm, i.p; twice a day for three days) treatment improved locomotor behavior in stroke rats[8]. Pifithrin-α reduced dopaminergic neurodegeneration in animal models of Parkinson s disease(10 µg)[9], prevented cell death of dopaminergic transplants in 6-OHDA-lesioned rats(2 mg/kg/day; i.p ; 5 days)[10], improved the survival of neuroprogenitor cells in subventricular zone of stroke rats, and reduced traumatic brain injury[11].
References:
[1]. Komarov PG, Komarova EA, et,al. A chemical inhibitor of p53 that protects mice from the side effects of cancer therapy. Science 285:1733-1737
[2]. Zhu X, Yu QS, et,al. Novel p53 inactivators with neuroprotective action: syntheses and pharmacological evaluation of 2-imino-2,3,4,5,6,7-hexahydrobenzothiazole and 2-imino-2,3,4,5,6,7-hexahydrobenzoxazole derivatives. J Med Chem 45:5090-5097
[3]. Hoagland MS, Hoagland EM, et,al. The p53 inhibitor pifithrin-alpha is a potent agonist of the aryl hydrocarbon receptor. J Pharmacol Exp Ther. 2005 Aug;314(2):603-10. doi: 10.1124/jpet.105.084186. Epub 2005 Apr 20. PMID: 15843497.
[4]. Chen YH, Bae E, et,al. Pifithrin-Alpha Reduces Methamphetamine Neurotoxicity in Cultured Dopaminergic Neurons. Neurotox Res. 2019 Aug;36(2):347-356. doi: 10.1007/s12640-019-00050-w. Epub 2019 May 8. PMID: 31069753.
[5]. Moretti D, Del Bello B, et,al. Calpain-3 impairs cell proliferation and stimulates oxidative stress-mediated cell death in melanoma cells. PLoS One. 2015 Feb 6;10(2):e0117258. doi: 10.1371/journal.pone.0117258. PMID: 25658320; PMCID: PMC4319969.
[6]. Guo J, Tang Q, et,al. Pifithrin-α enhancing anticancer effect of topotecan on p53-expressing cancer cells. Eur J Pharm Sci. 2019 Feb 1;128:61-72. doi: 10.1016/j.ejps.2018.11.024. Epub 2018 Nov 22. PMID: 30472223.
[7].Leker RR, Aharonowiz M, et,al. The role of p53-induced apoptosis in cerebral ischemia: effects of the p53 inhibitor pifithrin alpha. Exp Neurol. 2004 Jun;187(2):478-86. doi: 10.1016/j.expneurol.2004.01.030. PMID: 15144874.
[8]. Luo Y, Kuo CC, et,al. Delayed treatment with a p53 inhibitor enhances recovery in stroke brain. Ann Neurol. 2009 May;65(5):520-30. doi: 10.1002/ana.21592. PMID: 19475672; PMCID: PMC2690614.
[9]. Liang ZQ, Li YL, et,al. NF-kappaB contributes to 6-hydroxydopamine-induced apoptosis of nigral dopaminergic neurons through p53. Brain Res 1145:190-203
[10]. Chou J, Greig NH, et,al. Enhanced survival of dopaminergic neuronal transplants in hemi-Parkinsonian rats by the p53 inactivator PFT-α. Cell Transplant 20:1351-1359
[11]. Huang YN, Yang LY, et,al. Neuroprotective effects of pifithrin-α against traumatic brain injury in the striatum through suppression of neuroinflammation, oxidative stress, autophagy, and apoptosis. Sci Rep. 2018 Feb 5;8(1):2368. doi: 10.1038/s41598-018-19654-x. PMID: 29402897; PMCID: PMC5799311.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















