AMG-176 (Synonyms: AMG-176) |
| Catalog No.GC19452 |
AMG-176 (AMG-176) is a potent, selective and orally active MCL-1 inhibitor, with a Ki of 0.13 nM.
Products are for research use only. Not for human use. We do not sell to patients.
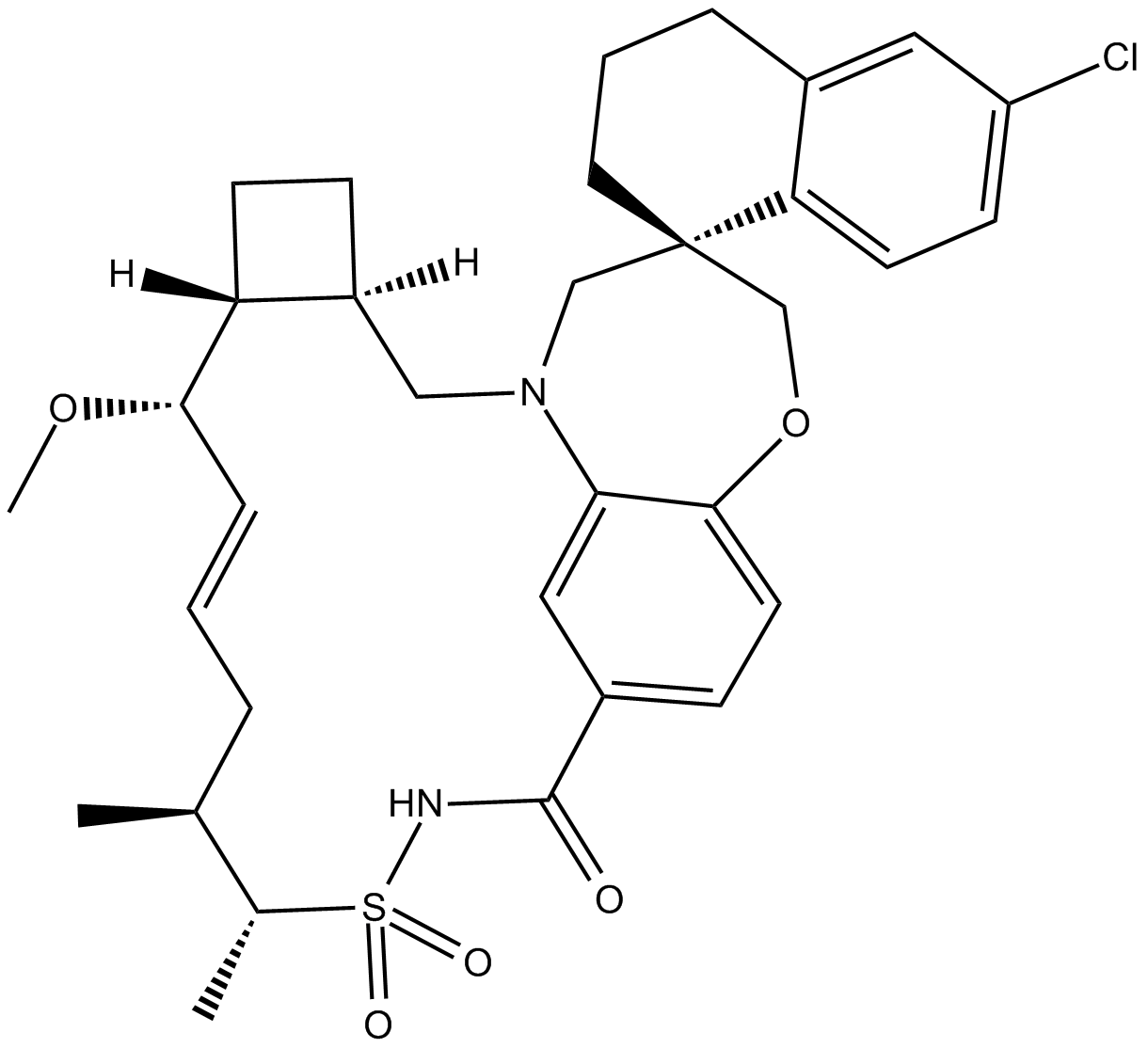
Cas No.: 1883727-34-1
Sample solution is provided at 25 µL, 10mM.
AMG-176 (Tapotoclax) is a potent, selective and orally active MCL-1 inhibitor, with a Ki of 0.13 nM. AMG-176 is a potent antagonist of Mcl-1; in cell free system the Ki value is <1 nM. In whole cells, the drug was tested in different leukemias, lymphoma, and multiple myeloma cell lines[1]. AMG-176 incubations resulted in time- and dose-dependent CLL cell death. At 100 and 300 nmol/L, there was 30% and 45% cell death at 24 hours. These concentrations did not result in significant cell death in normal hematopoietic cells. Presence of stroma did not affect AMG-176-induced CLL cell death[4].AMG-176 induced cell death was inversely correlated with endogenous McL-1 levels[5].
AMG-176 has a weak ability to induce apoptosis in C002M melanoma cell line, but a strong ability to induce apoptosis in C057M melanoma cell line. AMG-176 has a synergistic effect with I-BET151 in these two melanoma cell lines[2]. AMG-176 completely neutralized the survival effect of the IDO1/Kyn/AHR pathway in CLL cells[6].
AMG-176 Is Efficacious in Multiple Myeloma. In vivo, Dose-dependent activation of the intrinsic apoptosis pathway in OPM-2 xenografts, as measured by activated BAK, cleaved caspase-3, and cleaved PARP, was detected as early as 2 hours after oral administration of AMG 176, with sustained cleaved PARP and activated BAK detectable through 12 hours and cleaved caspase-3 through 24 hours. Immunohistochemistry analysis revealed a similar dose-dependent increase in cleaved caspase-3[3].
References:
[1]:Garner TP, Lopez A, et,al. Progress in targeting the BCL-2 family of proteins. Curr Opin Chem Biol. 2017 Aug;39:133-142. doi: 10.1016/j.cbpa.2017.06.014. Epub 2017 Aug 17. PMID: 28735187; PMCID: PMC5667545.
[2]: Tseng HY, Dreyer J, et,al.Co-targeting bromodomain and extra-terminal proteins and MCL1 induces synergistic cell death in melanoma. Int J Cancer. 2020 Oct 15;147(8):2176-2189. doi: 10.1002/ijc.33000. Epub 2020 Apr 24. PMID: 32249419.
[3]: Caenepeel S, Brown SP,et,al. AMG 176, a Selective MCL1 Inhibitor, Is Effective in Hematologic Cancer Models Alone and in Combination with Established Therapies. Cancer Discov. 2018 Dec;8(12):1582-1597. doi: 10.1158/2159-8290.CD-18-0387. Epub 2018 Sep 25. Erratum in: Cancer Discov. 2019 Jul;9(7):980. PMID: 30254093.
[4]:Yi X, Sarkar A, et,al. AMG-176, an Mcl-1 Antagonist, Shows Preclinical Efficacy in Chronic Lymphocytic Leukemia. Clin Cancer Res. 2020 Jul 15;26(14):3856-3867. doi: 10.1158/1078-0432.CCR-19-1397. Epub 2020 Jan 14. PMID: 31937611; PMCID: PMC7358119.
[5]: Yi X, Jain N, et,al. Targeting Mcl-1 by AMG-176 During Ibrutinib and Venetoclax Therapy in Chronic Lymphocytic Leukemia. Front Oncol. 2022 Feb 22;12:833714. doi: 10.3389/fonc.2022.833714. PMID: 35273915; PMCID: PMC8901605.
[6]: Atene CG, Fiorcari S, et,al. Indoleamine 2, 3-Dioxygenase 1 Mediates Survival Signals in Chronic Lymphocytic Leukemia via Kynurenine/Aryl Hydrocarbon Receptor-Mediated MCL1 Modulation. Front Immunol. 2022 Mar 18;13:832263. doi: 10.3389/fimmu.2022.832263. PMID: 35371054; PMCID: PMC8971515.
Average Rating: 5 (Based on Reviews and 10 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















