Valspodar (Synonyms: Valspodar) |
| Catalog No.GC16677 |
A multidrug-resistance modulator
Products are for research use only. Not for human use. We do not sell to patients.
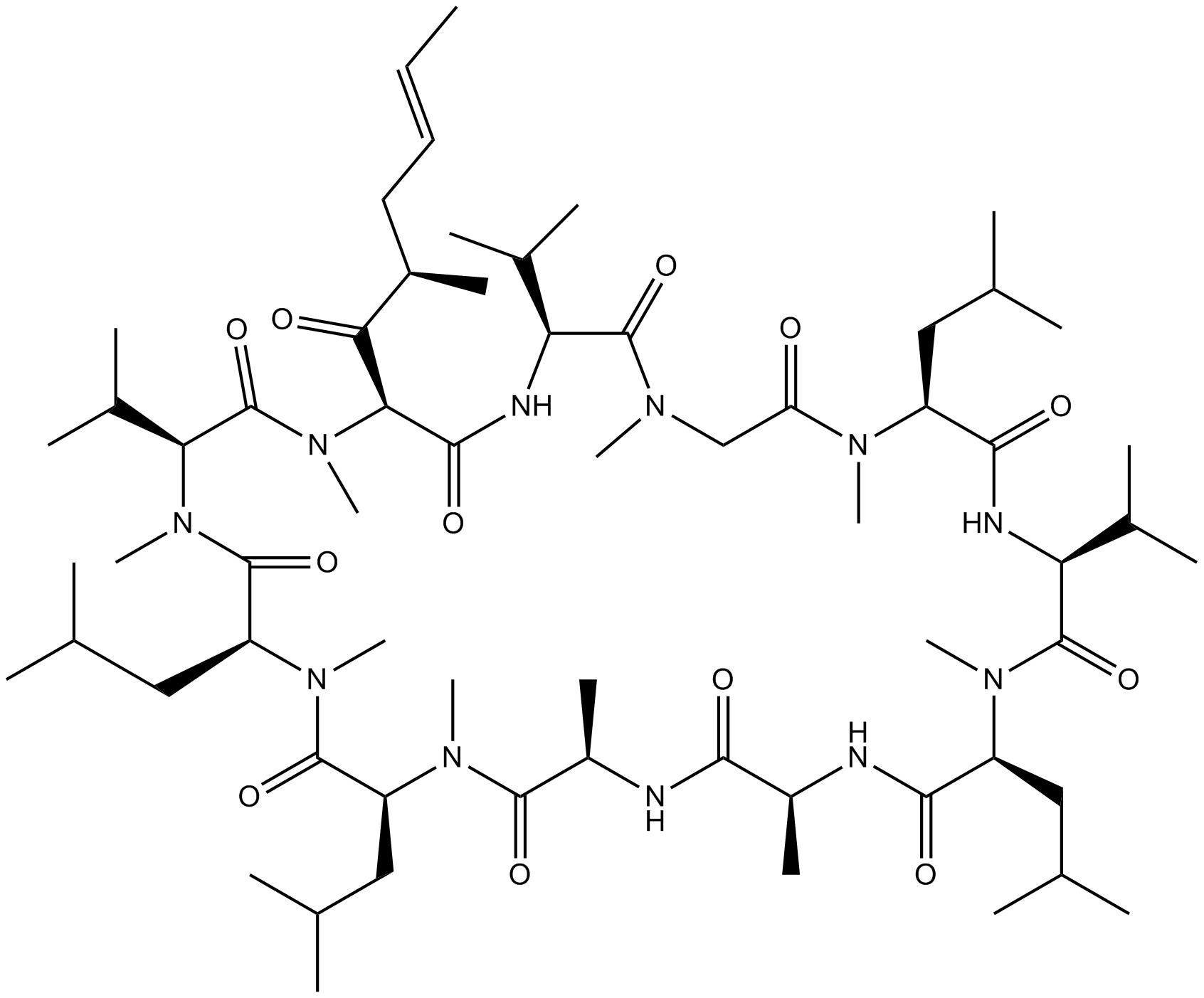
Cas No.: 121584-18-7
Sample solution is provided at 25 µL, 10mM.
Valspodar is a potent inhibitor of P-glycoprotein (P-gp) widely used in preclinical and clinical studies [1].
P-gp is a transmembrane glycoprotein which is located on cell membrane. P-gp distributes extensively and is expressed in certain cell types primarily containing liver, colon, kidney and pancreas. It also is known as multidrug resistance protein 1 (MDR1) which is pumps foreign substances out of cells. P-gp decreases the net uptake of cytotoxic drugs into the cells and mediats the efflux of these agents out of the cells, which is ATP-dependent. P-gp also overexpress in some cancer cells. P-gp plays an important role in mediating resistance to anticancer drugs and decreasing drug accumulation in multidrug-resistant cancer cells.[1]
Valspodar can reverse the resistance to mitoxantrane which is due to the expression of P-gp. The IC50 of mitoxantrane decreased from 1.6 ± 0.13 μM to 0.4 ± 0.02
μM in MDA-MB-435mdr cells pretreated with 3 mg/ml PSC. Valspodar increase the mitoxantrane intracellular accumulation by decreasing drug efflux and increasing mitoxantrone net uptake in cells.[1] The cytotoxicity was significant greater in T47D/TAMR-6 cells treated with doxorubicin and valspodar than doxorubicin only. Co-encapsulation of doxorubicin and valspodar presents a promising anticancer effect.[2] Valspodar was rapid absorpted and reachs the peak within 2 hnafter an oral dose. Valspodar showed properties of wide distribution, low hepatic extraction and mean bioavailability of 42.8% in rat.[3]
References:
[1]. Shen F, Bailey BJ, Chu S, Bence AK, Xue X, Erickson P, Safa AR, Beck WT, Erickson LC: Dynamic assessment of mitoxantrone resistance and modulation of multidrug resistance by valspodar (PSC833) in multidrug resistance human cancer cells. J Pharmacol Exp Ther 2009, 330(2):423-429.
[2]. Bajelan E, Haeri A, Vali AM, Ostad SN, Dadashzadeh S: Co-delivery of doxorubicin and PSC 833 (Valspodar) by stealth nanoliposomes for efficient overcoming of multidrug resistance. J Pharm Pharm Sci 2012, 15(4):568-582.
[3]. Binkhathlan Z, Hamdy DA, Brocks DR, Lavasanifar A: Pharmacokinetics of PSC 833 (valspodar) in its Cremophor EL formulation in rat. Xenobiotica 2010, 40(1):55-61.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















